|
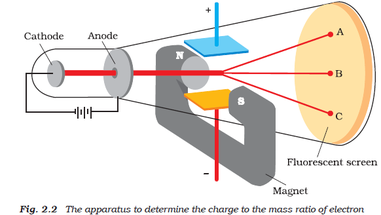
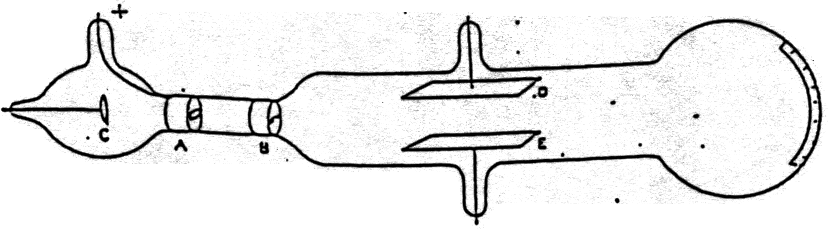
Thomson also concluded that the cathode rays were made up of negatively charged particles that he called electrons. Thomson concluded that the cathode rays were negatively charged and were being repelled by the metal plate. When the magnet was placed near the anode, the cathode rays were deflected away from the anode. When the magnet was placed near the cathode, the cathode rays were deflected towards the anode. Thomson used a magnet to deflect the cathode rays.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
